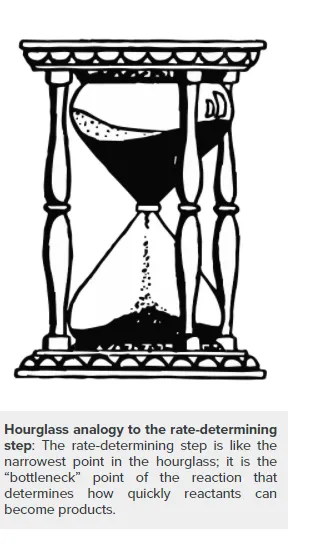
Rate-Determining Steps
The rate of a multi-step reaction is determined by the slowest elementary step, which is known as the rate-determining step.
Chemists often write chemical equations for reactions as a single step that shows only the net result of a reaction. However, most chemical reactions occur over a series of elementary reactions. The complete sequence of these elementary steps is called a reaction mechanism. The reaction mechanism is the step-by-step process by which reactants actually become products. It is the “how” of the reaction, whereas the overall balanced equation shows only the “what” of the reaction. In kinetics, the rate of a reaction with several steps is determined by the slowest step, which is known as the rate-determining, or rate-limiting, step.
Take the following example of a gas phase reaction:


