Diffusion
Diffusion Mechanisms
Diffusion is the process of mass flow in which atoms change their positions relative to neighbors in a given phase under the influence of thermal and a gradient. The gradient can be a compositional gradient, an electric or magnetic gradient, or stress gradient. Many reactions in solids and liquids are diffusion dependent. Diffusion is very important in many industrial and domestic applications. E.g.: Carburizing the steel, annealing homogenization after solidification, coffee mixing, etc. From an atomic perceptive, diffusion is a step wise migration of atoms from one lattice position to another. Migration of atoms in metals/alloys can occur in many ways, and thus corresponding diffusion mechanism is defined.
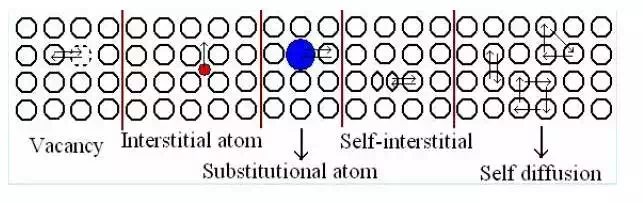
Atom diffusion can occur by the motion of vacancies (vacancy diffusion) or impurities (impurity diffusion). The energy barrier is that due to nearby atoms which need to move to let the atoms go by. This is more easily achieved when the atoms vibrate strongly, that is, at high temperatures. There is a difference between diffusion and net diffusion. In a homogeneous material, atoms also diffuse but this motion is hard to detect. This is because atoms move randomly and there will be an equal number of atoms moving in one direction than in another. In inhomogeneous materials, the effect of diffusion is readily seen by a change in concentration with time. In this case there is a net diffusion. Net diffusion occurs because, although all atoms are moving randomly, there are more atoms moving in regions where their concentration is higher.