Structure of Solids
Structure of Solids - All solid substances are either amorphous solids or crystalline solids. In the amorphous solids, the atoms are arranged chaotically, i.e., the atoms are not arranged in a systematic order. The common amorphous solids are wood, plastics, glass, paper, rubber etc. In crystalline solids, the atoms making up the crystals arrange themselves in a definite and orderly manner and form. All solid metals such as iron, copper, aluminium etc. are crystalline solids. The definite and orderly manner and form of atoms producing a geometrical shape in the aggregate is called space lattice or crystal lattice.
A crystal is composed of unit cells. A unit cell contains the smallest number of atoms, which when taken together have all the properties of the crystals of the particular metal. The unit cells are arranged like building blocks in a crystal, i.e. they have the same orientation and their similar faces are parallel. A unit cell may also be defined as the smallest parallelopiped which could be transposed in three coordinate directions to build the space lattice. The space lattice of various substances differ in size and shape of their unit cells.
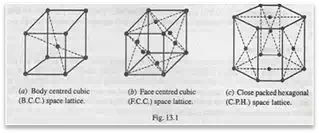
According to Bravais (a scientist), there are fourteen possible types of space lattices, but the following three types are usually found in most of the metals.
1. Body centred cubic (B.C.C.) space lattice. In a unit cell of body centred cubic space lattice, there are nine atoms. The eight atoms are located at the corners of the cube and one atom at its centre, as shown in Fig. 13.1 (a). This type of lattice is found in alpha iron, tungsten, chromium manganese, molybdenum, tantalum, barium, vanadium etc.
2. Face centred cubic (F.C.C.) space lattice. In a unit cell of face centred cubic space lattice, there are fourteen atoms. The eight atoms are located at the corner of the cube and six atoms at the centres of six faces, as shown in Fig. 13.1 (b). This type of lattice is found in gamma iron, aluminium, copper, lead, silver, nickel, gold, platinum, calcium etc.
3. Close packed hexagonal (C.P.H.) space lattice. In a unit cell of close packed hexagonal space lattice, there are seventeen atoms. The twelve atoms are located at the twelve corners of the hexagonal prism, one atom at the centre of each of the two hexagonal faces and three atoms are symmetrically arranged in the body of the cell as shown in Fig. 13.1 (c). This type of lattice is found in zinc, magnesium, cobalt, cadmium, antimony, bismuth, beryllium, titanium, zirconium etc.