
Deaeration in boilers
In order to meet industrial standards for both oxygen content and the allowable metal oxide levels in feed water, nearly complete oxygen removal is required. This can be accomplished only by efficient mechanical deaeration supplemented by a properly controlled oxygen scavenger.
Deaeration is driven by the following principles: the solubility of any gas in a liquid is directly proportional to the partial pressure of the gas at the liquid surface, decreases with increasing liquid temperature; efficiency of removal is increased when the liquid and gas are thoroughly mixed.
Deaeration can be performed using a physical medium such as deaerating heaters or vacuum deaerators or a chemical medium such as oxygen scavengers (polishing treatment) or catalytic resins. Membrane contractors are increasingly being used. Carbon dioxide is often removed using a physical medium.
The purpose of a deaerator is to reduce dissolved gases, particularly oxygen, to a low level and improve plant thermal efficiency by raising the water temperature. In addition, they provide feed water storage and proper suction conditions for boiler feed water pumps.
Pressure deaerators can be classified under two major categories: tray type and spray type.
The tray type desecrating heaters consist of a shell, spray nozzles to distribute and spray the water, a direct contact vent condenser, tray stacks and protective interchamber walls. The chamber is constructed in low carbon steel, but more corrosion-resistant stainless steels are used for the spray nozzles and the other parts.
Incoming water is sprayed into steam atmosphere, where it is heated up to a few degrees to the saturation temperature of the steam. Most of the non-condensable gases (principally oxygen and free carbon dioxide) are released to the steam as the water is sprayed into the unit. Seals prevent the recontamination of tray stack water by gases from the spray section. Water falls from tray to tray, breaking into fine droplets of film, which intimately contact the incoming steam.

The steam heats the water to the steam saturation temperature and removes the very last traces of oxygen. Deaerated water falls to the storage space below, where a steam blanket protects it from recontamination. It is usually stored in a separate tank.
The steam enters the deaerators through ports in the tray compartment, flows down through the tray stack parallel to the water flow. A very small amount of steam condenses in this section as the water temperature rises to the saturation temperature of the steam. The rest of the steam scrubs the cascading water. Before leaving the tray compartment, the steam flows upward between the shell and the interchamber walls to the spray section. Most of the steam is condensed and becomes part of the deaerated water. A small portion of the steam, which contains the non-condensable gas released from the water, is vented to the atmosphere. It is essential that sufficient venting is provided at all times or deaeration will be incomplete. Steam flow through the tray stack may be cross-flow, counter-current, or co-current to the water.
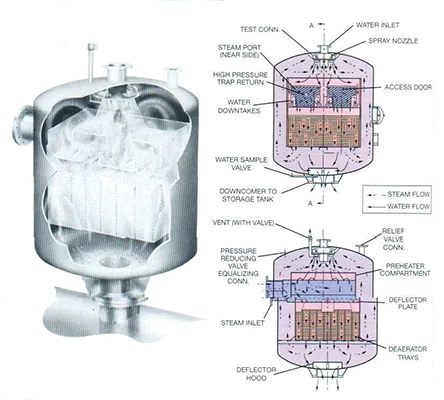
The spray type deaerating heaters consist of a shell, spring-loaded inlet spray valves, a direct contact vent condenser section and a steam scrubber for final dearetion; the shell and steam may be low carbon steel, the spray valves and the direct contact vent condenser section are in stainless steel. The incoming water is sprayed into a steam atmosphere and heated up to a few degrees to the saturation temperature of the steam. Most of the non-condensable gases are released to the steam, and the heated water falls to water seals and drains to the lowest section of the steam scrubber. The water is scrubbed by a large volume of steam and heated to the saturation temperature prevailing at that point. As the water-steam mixture rises in the scrubber, the deaerated water is a few degrees above the saturation temperature, due to a slight pressure loss. In this way a small amount of flashing is produced, which aids in the release of dissolved gases. The deaerated water overflows from the steam scrubber to the storage section below.
Steam enters the deaerator through a chest on the side and flows to the steam scrubber. After flowing into the scrubber it passes up into the spray heater section to heat the incoming water. Most of the steam condenses in the spray section to become a part of the deaerated water. A small portion of the gases is vented to the atmosphere to remove the non-condensable gases.
Vacuum deaeration is used at temperatures below the atmospheric boiling point to reduce the corrosion rate in water distribution systems. A vacuum is applied to the system to bring the water to its saturation temperature. Spray nozzles break the water into small particles to facilitate gas removal and vent the exhaust gases. Incoming water enters through spray nozzles and falls through a columns packed with Raschig rings to other synthetic packing. In this way, water is reduced to thin films and droplets, which promote the release of dissolved gases. The released gases and water vapor are removed through the vacuum, which is maintained by steam jet eductors or vacuum pumps, depending on the size of the system. Vacuum deaerators remove oxygen less efficiently that pressure units.
Corrosion fatigue at or near welds is a major problem in deaerators. It is the result of mechanical factors, such as manufacturing procedures, poor welds and lack of stress-relieved welds. Operational problems such as water/steam hammer can also be a factor.