Chemistry
For the difluoromethylation reaction, the inlets A, B and D were attached with standard connectors to three syringe pumps (Asia Syrris). Inlet C was connected via a calibrated mass flow controller (Bronkhorst) to the fluoroform gas cylinder, and the outlet was connected to a backpressure regulator (Swagelok 0–25 bar). Finally, the cooling element was attached in a parallel flow arrangement to the circulation cryostat (Fig. 7). Diphenylacetonitrile was used a model substrate for the difluoromethylation reaction to demonstrate the performance of the printed reactor. The reaction conditions for the flow difluoromethylation were chosen to resemble those reported by Mikami and co-workers.18 A comprehensive re-optimization of the reaction conditions was not attempted at this stage. Thus, the reactor was cooled by the cryostat to a temperature of −65 °C. A 0.5 M solution of diphenylacetonitrile was pumped into Inlet A with a flow rate of 0.8 mL min−1, while a commercial solution of nBuLi in hexane (2.5 M) was pumped directly from the supply bottle into Inlet B with a flow rate of 0.36 mL min−1(Fig. 7). With these flow rates a 2.25 fold excess of nBuLi and a nominal residence time of ∼50 s in the first reaction zone were obtained. After the first reaction zone the mixture was combined with the fluoroform gas at a flow rate of 26.7 mL min−1 (flow rate at standard conditions; 3 equivalents). Preceding experiments with CHF3 in continuous flow reactors made of Teflon tubings demonstrated that the gas is completely dissolved in the liquid phase at this temperature. In the second zone of the reactor, the fluoroform is deprotonated by the remaining nBuLi and the resulting unstable trifluoromethanide anion subsequently loses a fluoride ion in an α-elimination to generate a short-lived singlet difluorocarbene (CF3− → :CF2 + F−). The electrophilic singlet difluorocarbene then reacts with the deprotonated substrate (Fig. 7). The nominal residence time in the second reactor zone is ∼1 min. At Inlet D, the reaction mixture is finally combined with a quench solution of MeOH at a flow rate of 1 mL min−1 before it left the reactor through the outlet and a backpressure regulator at 5 bar. The product was formed with 95% selectivity in this reaction according to GC-FID (see Experimental section for details). The collected mixture was then extracted with Et2O/H2O, the solvent evaporated and the crude product finally recrystallized from n-hexane. 4 mL of starting material were processed in a continuous run to afford 395 mg of the desired difluoromethylated product in excellent purity and 81% yield.
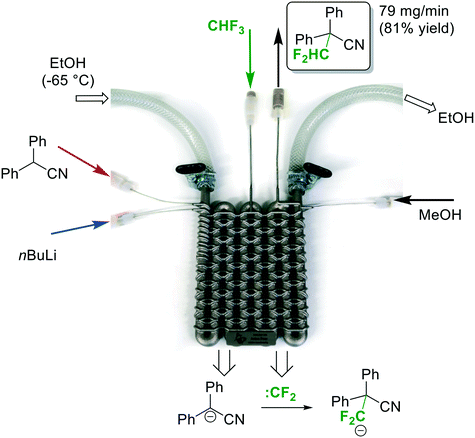 Fig. 7 Continuous flow Cα-difluoromethylation
with fluoroform.
Fig. 7 Continuous flow Cα-difluoromethylation
with fluoroform.
Conclusion
With the development of new materials for additive manufacturing, such as various metals, alloys, and ceramics, and improving accuracy and quality of the outputs, the production of customized, functional end-products has become increasingly feasible. Even though these technologies are still rarely applied in organic synthesis, recent examples demonstrate that chemical reactors of almost arbitrary complexity can be produced.7–9,12,13 Herein we presented the design and 3D printing of a continuous flow reactor by selective laser melting (SLM) of stainless steel powder. The utilization of stainless steel enables fast heat transfer and high pressure resistance, thus, allowing the execution of exothermic reactions at cryogenic temperatures and elevated pressures. Furthermore, reactors printed from stainless steel allow the utilization of reagents/solvents which would be destructive to the majority of materials commonly used for other rapid prototyping techniques (such as PDMS). The reactor was specifically designed for a two-step difluoromethylation reaction with gaseous fluoroform. This reaction produces the desired product in excellent purity after a total reaction time of less than 2 min at a reaction temperature of −65 °C. It can be expected that additive manufacturing and related direct digital manufacturing technologies, in combination with computational chemical reaction and fluid dynamics simulation, will play a fundamental role in the design of next-generation, continuous flow microreactors. Further work is planned in our laboratories to integrate in-line analysis into the reactor and to further explore difluroromethylation chemistry with fluoroform and other multiphase (g/l) reactions.